Dr David Stephens Introduces a Consensus Guideline to Assist Primary Care in the Assessment, Management, and Treatment of Osteoporosis
| Read This Article to Learn More About: |
|---|
Find take-home messages for GPs, guidance during the COVID-19 pandemic, and implementation actions for STPs, ICSs, and clinical pharmacists in general practice at the end of this article |
Over 3 million people in the UK are estimated to have osteoporosis1 and every year there are around 500,000 fragility fractures, leading to social isolation, loss of independence, disability, long-term pain, and premature death.1 Reduced bone density is the major risk factor for fragility fracture, but other factors also increase the risk. Increasing age in both sexes and menopause in women lead to increased bone loss, and therefore the prevalence of osteoporosis increases markedly with age—for example, in women it increases from 2% at age 50 years to more than 25% at age 80 years.2
Identifying patients at very high risk of fragility fractures is likely to decrease the severe mortality (20%)2 and morbidity from hip and other fragility fractures.
Introduction
Three different UK guidelines for osteoporosis have been published over the last 10 years2–4 and even for specialists, this can be confusing. During 2020, a working party group convened to produce a new UK osteoporosis guideline5 to supplement this other guidance and to endorse the revolution in the treatment of patients at very high risk, and some at high risk, of osteoporosis fracture.5
The UK consensus guideline on the management of patients at low, high, and very high risk of osteoporotic fracture5 was written by a team of bone and primary care specialists who have reviewed the latest evidence. It is intended to support primary care in the management of people at different risks of osteoporotic fracture. Its recommendations are based on evidence from studies of women after the menopause, although they can be used in men with osteoporosis. This article aims to explain this new guideline and how it supplements other UK guidance. Throughout the rest of this article the guideline is referred to as the consensus guideline. A summary algorithm from the consensus guideline is shown in Figure 1.5
Note: Not all of the treatments discussed in this article currently (December 2020) have UK marketing authorisation. The prescriber should follow relevant professional guidance, taking full responsibility for all clinical decisions. Informed consent should be obtained and documented. See the General Medical Council’s guidance on Good practice in prescribing and managing medicines and devices6 for further information.
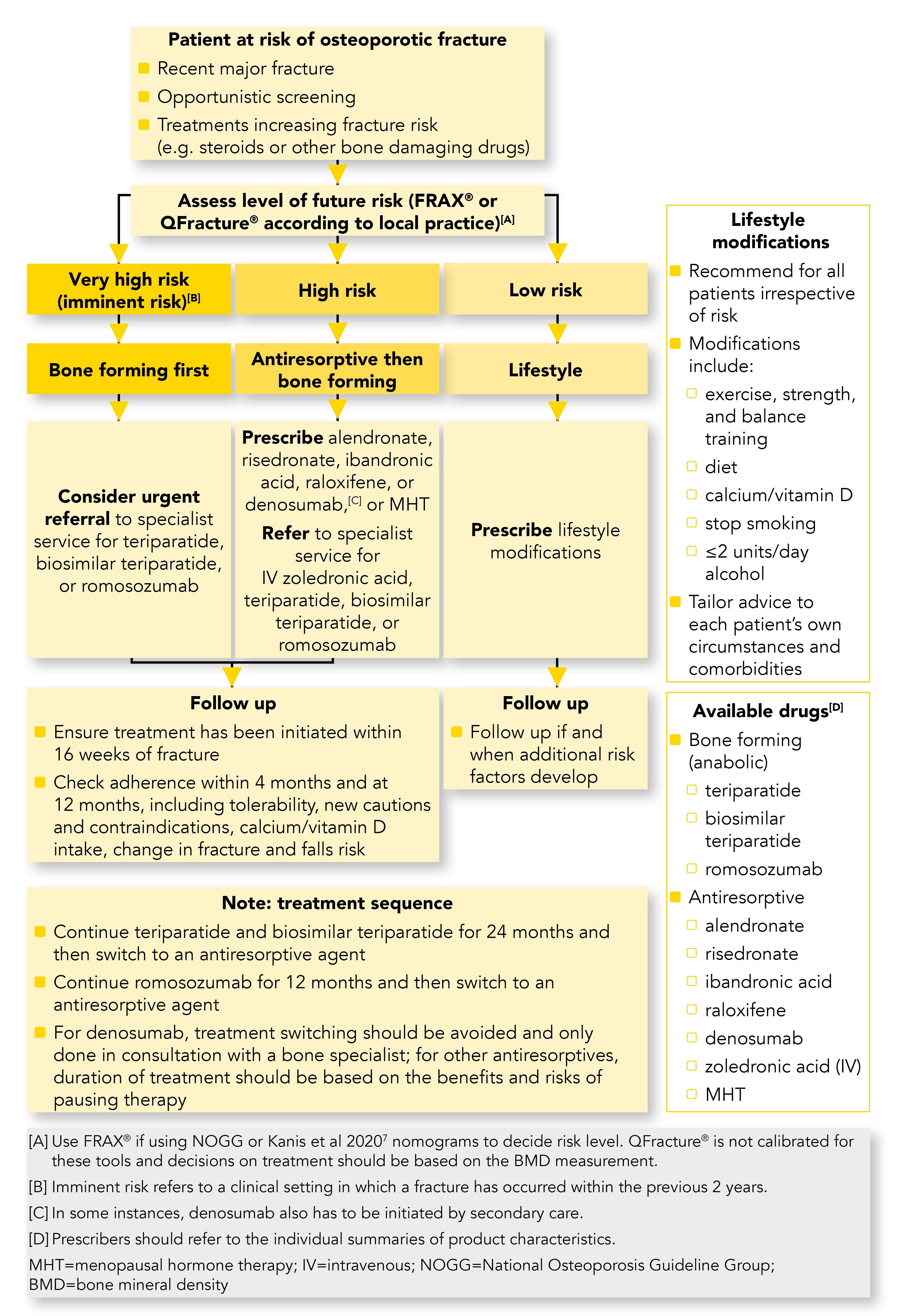
Managing Patients Based on Their Future Risk
Although bone-building medications and antiresorptive treatments have clearly been shown to reduce the risk of a further fracture, individual patient factors should be considered when deciding on treatment options.5 For example, people with serious illness such as advanced cancer or advanced dementia may not benefit from treatment, while a younger patient with a lower risk but who smokes, drinks too much alcohol, and is inactive may benefit from treatment lasting many years.
Lifestyle Modification
Lifestyle modification should be recommended for all patients irrespective of their level of risk (see Box 1).5
The consensus guideline recommends exercise for all, adequate dietary calcium and vitamin D, falls prevention, cessation of smoking, and a maximum of 14 units of alcohol per week. Postural care is recommended for people with vertebral fractures.
| Box 1: Lifestyle Modifications to Improve Bone Health5 |
|---|
|
Patients at Very High Risk
In patients with osteoporosis who are at very high risk, the consensus guideline recommends prompt intravenous or injectable bone-building treatments to prevent a further fracture.5 The evidence indicates that after a fragility fracture, instead of patients having treatment with oral alendronate, they should be considered for urgent referral for treatment with one of the anabolic bone-building medications—teriparatide, biosimilar parathyroid hormone, or romosozumab. These treatments are usually delivered by a bone specialist. Antiresorptive therapy is subsequently given to maintain the effect. A more rapid and greater fracture risk reduction with anabolic treatments compared with antiresorptive treatments has been demonstrated, particularly in people at very high risk of fractures, and the effect of these agents on bone mineral density (BMD) can be maintained with bone-turnover inhibitors once anabolic treatment has stopped.7
Patients at High Risk
Patients at high risk of future fracture should be treated with antiresorptive medication before referral for bone-forming drugs. Prescribe alendronate, risedronate, ibandronic acid, raloxifene, denosumab, or hormone replacement therapy, or refer to specialist services for intravenous zoledronic acid.5
Both alendronate and zoledronic acid reduce the refracture rate significantly and while alendronate is oral, zoledronic acid is given by an intravenous infusion. Zoledronic acid infusions are only required in a single dose once a year, for 3 years,10 potentially addressing the major issue of concordance, which has dogged oral resorptive treatments ever since they were introduced.
If antiresorptive medications have been tried and are not effective, due to either an adverse reaction or continuing deterioration in the bone density, then refer the patient to specialist services for bone-forming (anabolic) treatment with teriparatide, biosimilar teriparatide, or romosozumab.5
Patients at Low Risk
Patients who are at low risk should be reassured and given advice on lifestyle modifications (Box 1), calcium and vitamin D nutrition, hormone treatment in perimenopausal and postmenopausal in women, and testosterone replacement therapy in men with male hypogonadism.5
How to Assess Risk
The consensus guideline identifies the three presentations of osteoporosis: a recent (within the last 2 years) major fracture, opportunistic screening, and treatments that increase fracture risk (for example, steroids or other bone-damaging drugs).5
It includes the following definition of osteoporosis:3,5
‘People with a T-score that is 2.5 standard deviations or more below the young adult mean value for women are diagnosed with osteoporosis, and if in addition to this there are one or more documented fragility fractures they are diagnosed with severe or established osteoporosis.’
Some patients can be diagnosed with osteoporosis without the need for measurement of BMD, for example, after specific fragility fractures and by age.5
All patients with a prior fragility fracture are at least high risk and possibly very high risk depending on their FRAX® probability.5,9
Very High, High, and Low Risk Rategories
Professor Kanis and his team, who developed FRAX®, have published an article on the assessment and specific treatment of women that stratifies patients at risk of osteoporosis into very high, high, and low risk groups.9 FRAX® is a computerised risk tool that calculates the risk of a fragility fracture in an individual over the next 10 years. Using FRAX®, Kanis divided patients into these risk groups based on age and the 10-year probability of a further fracture and recommended prompt treatment for people in the very high risk group.9
Kanis defines people in the very high risk group (also sometimes termed the ‘imminent risk group’, see Figure 1) as patients with a high FRAX® score and a history of major fracture of the hip/pelvis/femur, vertebra, humerus, or ribs in the last 2 years. These patients have a fracture probability that lies above the upper assessment threshold after a FRAX® risk calculation. Risk can be calculated using FRAX® with or without the inclusion of BMD, so that where BMD testing is unavailable, the same probability threshold can be used.9 Measuring BMD using dual-energy X-ray absorptiometry (DXA) is the gold standard for the diagnosis of osteoporosis. The DXA result can be incorporated into FRAX® to increase the accuracy of the risk calculation. It is worth noting that patients should always have their risk recalculated after a fragility fracture.
The high risk category is divided by Kanis into those falling above his recommended intervention threshold and those below it.9 Patients whose initial risk assessment with FRAX®, using clinical factors alone, falls between the upper and lower assessment thresholds should have this initial FRAX® assessment followed by BMD assessment and recalculation of FRAX® probability including femoral neck BMD. The patient may then be reclassified; those reclassified as being at very high or at low risk are treated as described above, while those who remain above the ‘high risk’ intervention threshold but below the ‘very high risk’ assessment threshold are considered to be at high risk, and initial antiresorptive therapy should be considered.9 Those whose risk falls below the intervention threshold after BMD reassessment are shown to be ‘low risk’.
Patients older than the age limits for the tools (90 years for FRAX® and 99 years for QFracture®) should be considered at high risk (QFracture® is an alternative risk calculator).2
Key Medications
The consensus guideline notes the bone-forming anabolic medications teriparatide, biosimilar teriparatide, and the new monoclonal romosozumab. In the guideline, romosozumab has been added to the pharmaceutical interventions for patients at very high and high risk. See Box 2 for a summary of the drugs available to treat osteoporosis.5 Teriparatide, which in the 2017 NOGG guidance was second-line treatment and reserved for patients at very high risk, is now first-line treatment for patients at very high risk (Figure 1).3,5,9
| Box 2: Drugs Available to Treat OsteoporosisA,5 |
|---|
Note A: Prescribers should refer to the individual summaries of product characteristics.11–16,19–22 IV=intravenous |
The consensus guideline confirms the use of antiresorptive medications, that is, the bisphosphonates: alendronate, risedronate, ibandronic acid, and zoledronic acid. Raloxifene, a selective oestrogen receptor modulator, can be used in women, as well as hormone replacement for men and women. The monoclonal antibody denosumab can also be used.5
Duration of Treatment
The duration and progression of treatments are reviewed, with a recommendation to continue with antiresorptive agents after 2 years of teriparatide, 1 year of romosozumab, and if stopping denosumab (only on specialist advice) to also continue with an antiresorptive. Switching from denosumab should be avoided and specialist advice should be sought.5
With regard to duration of treatment, the consensus guideline states that this should be as per local or national guidelines with review of the risks versus benefits of discontinuing therapy. It cites SIGN guidance that: alendronate and strontium can be continued for 10 years (if osteoporosis is severe and other treatments are unsuitable), risedronate for 7 years, and zoledronic acid for 3 years, although this can be used every 18 months for 6 years off licence. See Figure 1.4,5
Implementation Challenges
There are organisational and resource challenges to arrange treatment in line with the consensus guidance. These include:
- how does secondary care identify patients who are at imminent and high risk?
- how do GPs, having identified patients at imminent and high risk, facilitate prompt treatment?
The first point relates to where patients have had a fragility fracture and secondary prevention has not been initiated in secondary care, and the second to where patients have only received care in primary care for their fracture. The author’s personal experience, working in Scotland and also having worked in rural New Zealand, is that fragility fractures may often be treated in primary care.
The consensus guideline acknowledges the need for seamless care from secondary to primary care; on discharge from hospital, patients need to be assessed in primary care by a pharmacist, GP, or maybe a bone health lead in the practice.5 At this assessment the need for intravenous therapy can be reassessed. Perhaps there is now an enhanced role for Fracture Liaison Services in supporting or providing these elements of care.23
How the Consensus Guideline Fits in With Other UK Guidelines
Other current UK osteoporosis guidelines are SIGN 2015, updated in June 2020;4 NOGG 2017;3 and NICE 2012, updated 20172, whose guidance was on the assessment of the risk of fragility fracture with a separate technology appraisal (TA) on the medication.24
Both FRAX® and QFracture® intervention thresholds are based on expert recommendation rather than evidence. NICE declines to recommend intervention thresholds in its guidance on Osteoporosis: assessing the risk of fragility fracture.25 SIGN prefers the use of QFracture® for initially assessing the risk of fragility fracture, as FRAX® has not been available for independent verification.4 QFracture® does not define risk categories, so SIGN has an expert recommendation of a 10% risk threshold, and for treatment to be a professional decision. SIGN therefore did not categorise into very high, high, and low risk. However, SIGN recommends anabolic treatments in some patients at higher risk; teriparatide (parathyroid hormone 1-34) is recommended to prevent vertebral and non-vertebral fractures in postmenopausal women with severe osteoporosis. In addition, in postmenopausal women with at least two moderate or one severe low-trauma vertebral fractures, teriparatide is recommended over oral bisphosphonates, to prevent vertebral fracture.4 SIGN has also introduced treatment with zoledronic acid for osteopenia (that is a T-score of –1.0 to –2.5) in people aged 65 years and over.4
NICE recommends either FRAX® or QFracture® for the initial assessment before a DXA scan.2 NICE re-issued TA464 on Bisphosphonates for treating osteoporosis, in April 2019.24 This was because a TA should be an economic assessment and it was, in effect, a clinical assessment.26 NICE concluded the technology assessment by saying:24
‘The choice of treatment should be made on an individual basis after discussion between the responsible clinician and the patient, or their carers, about the advantages and disadvantages of the treatments available. If generic products are available, start treatment with the least expensive formulation, taking into account administration costs, the dose needed and the cost per dose.’
In effect, the lack of good evidence of intervention thresholds leaves clinicians to either make their own decisions (SIGN and NICE)2,4 or to go with the expert recommendations from Kanis’ paper,9 the NOGG guideline,3 and the recent consensus guideline where FRAX® is used to refine characterisation of risk.5
Summary
There is a broad spectrum of guidance on osteoporosis—SIGN takes a professional/patient-led approach, where the clinician, along with the patient, looks at the QFracture® and DXA scan risks and where there is a 10% fracture risk decides on treatment; NICE allows either QFracture® or FRAX® to aid decision-making but is unable currently to recommend intervention thresholds; and NOGG and the consensus guideline identify patients who are at imminent and high risk and treat on this basis.
As a GP in Scotland, I should adhere to SIGN guidance, but if I were to need further guidance on the identification of patients at very high risk I would need to go with FRAX®. It is hoped that the new consensus guideline will help practitioners manage patients at different levels of risk and guide appropriate treatment decisions.
Dr David Stephens
GP, Scotland
Member of the consensus guideline development group
| Take-home Messages for GPs |
|---|
|
| Guidance During the COVID-19 Pandemic5 |
|---|
BMD=bone mineral density; DXA=dual-energy X-ray absorptiometry; ROS=Royal Osteoporosis Society; IOF=International Osteoporosis Foundation |
| Implementation Actions for STPs and ICSs |
|---|
Written by Dr David Jenner, GP, Cullompton, Devon The following implementation actions are designed to support STPs and ICSs with the challenges involved with implementing new guidance at a system level. Our aim is to help you consider how to deliver improvements to healthcare within the available resources.
STP=sustainability and transformation partnership; ICS=integrated care system |
| Implementation Actions for Clinical Pharmacists in General Practice |
|---|
Written by Nicola Cree, Pharmaceutical Services Manager, Soar Beyond Ltd The following implementation actions are designed to support clinical pharmacists in general practice with implementing the guidance at a practice level. Clinical practice pharmacists can enhance and improve patient outcomes. Any such work will also support the PCN DES, which lists patients who have had a recent fall as prime targets for structured medication reviews.[A]
Guidance and training for service development is available for a number of different clinical areas from Soar Beyond. If you have clinical pharmacists in your practice or organisation, contact Soar Beyond to see how we can support with their clinical delivery, training and development soarbeyond.co.uk [A] NHS England and NHS Improvement. Network Contract Directed Enhanced Service Contract specification 2020/21—PCN requirements and entitlements. NHSEI, March 2020. Available at: www.england.nhs.uk/wp-content/uploads/2020/03/network-contract-des-specification-pcn-requirements-entitlements-2020-21.pdf PCN=Primary Care Network; DES=Direct Enhanced Service |

