Dr Fiona Collier provides an overview of the identification, assessment, and management of hidradenitis suppurativa in primary care, in line with the latest BAD guideline
| Read This Article to Learn More About: |
|---|
Find key points and implementation actions for STPs and ICSs at the end of this article |
For many years, hidradenitis suppurativa (HS) has been considered a rare disease that is largely unknown by GPs and nurses working in primary care and usually seen at an advanced stage by dermatologists and some surgical specialties. It has now been unmasked as a relatively common chronic inflammatory skin disease. Recognition of HS at an earlier, milder stage has raised the possibility of preventing its progression to the severe, scarring, life-altering disease that occurs in some patients. In addition, recent evidence has shown increased risk of diabetes, cardiovascular disease, depression, and premature death in people with HS. The British Association of Dermatologists (BAD) guideline on hidradenitis suppurativa, published in 2018, aims to provide guidance on management of the condition based on current evidence and expert opinion.1 This article highlights the key recommendations from the BAD HS guideline for implementation in primary care.
Note: not all of the treatments discussed in this article currently (August 2019) have UK marketing authorisation for the indications mentioned. The prescriber should follow relevant professional guidance, taking full responsibility for all clinical decisions. Informed consent should be obtained and documented. See the General Medical Council’s guidance on Good practice in prescribing and managing medicines and devices2 for further information.
Background
Hidradenitis suppurativa is defined as a chronic inflammatory skin disease characterised by painful, inflamed nodules of the skin that recur and persist at specific sites, often progressing to chronic purulent discharge, sinus formation, and scarring.3 Prevalence of HS is more than two times greater in women than in men, and is rarely seen before puberty; most of those affected are young to middle-aged adults.4 The disease frequently persists throughout adult life, sometimes becoming less severe in women after menopause. No accurate data is available for the UK, but studies in other European populations have shown a prevalence of 1–4%.5,6 HS is two to three times more common in African American and biracial people compared with those who have white skin.4
Causes of Hidradenitis Suppurativa
Current evidence suggests that a number of factors lead to the development of HS, and different patterns of the disease may indicate individual variations in the role of these factors. Around 35% of patients with HS have a family history of the condition, and genetic studies have identified specific gene mutations associated with HS in affected families.7 In cases of chronic inflammatory response within the skin that is not initiated by infection, abnormal immunomodulation appears to be the underlying cause but secondary bacterial infection may play a part in perpetuating inflammation.3
Lifestyle Factors
Hidradenitis suppurativa is more common in people who are current smokers. There is also a strong association with obesity, with women showing worsening severity of HS with increasing obesity.5,8 Smoking and obesity both have pro-inflammatory effects on the body, and obesity also causes increased skinfold friction and hormonal imbalance, which are likely to worsen the effects of HS. Hormonal factors are also believed to play a part in HS, particularly in women because of the peak of the disease in the reproductive years and the improvement sometimes seen following treatment with anti-androgen drugs.9
Co-morbidities and Associated Conditions
An important feature of HS are the co-morbidities experienced by many patients. People with HS have significantly increased risks of adverse cardiovascular outcomes and all-cause mortality, independent of confounders such smoking and socio-economic status, and nearly twice the risk of cardiovascular-associated death compared with controls.10 Diabetes is also more common in HS, along with dyslipidaemia, hypertension, and other features of the metabolic syndrome, and the association is strongest in those aged under 50 years, in both obese and non-obese patients.11
There is an association between HS and acne, and a history of pilonidal sinus is common and may be another manifestation of HS.12 A number of other conditions including anaemia, thyroid disorders, spondyloarthropathy, Crohn’s disease, and polycystic ovarian syndrome, all have an increased incidence in patients with HS. Squamous cell carcinoma occurring in chronic lesions is a rare but recognised complication.13 As with many chronic diseases, HS is associated with a higher rate of depression and anxiety, and its profound effect on quality of life is reflected in an increased risk of completed suicide.14,15
Diagnosis
Diagnosis of HS is clinical, and there are no diagnostic tests to detect it. Diagnosis is based on the triad of typical lesions at characteristic sites, with a recurrent or chronic pattern (Box 1). Patients with a characteristic clinical picture can be diagnosed confidently in primary care (see Figure 1).
Patients have often had symptoms for several years before receiving a diagnosis. They have usually had multiple short courses of antibiotics for treatment of abscesses and may also have had several acute lesions (often in the breast, axillae, and groin) incised and drained.
| Box 1: Triad of Features in Hidradenitis Suppurativa1 |
|---|
|
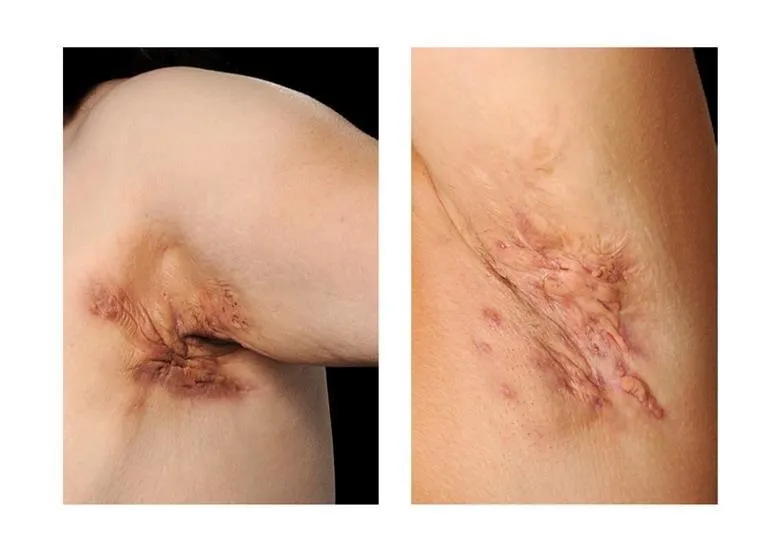
Source: Cardiff University Image Library, courtesy of Dr John Ingram. Reproduced with permission.
Initial Assessment
Initial assessment should include enquiries about which body sites are affected, the number of new and persisting HS lesions in the last month, symptoms such pain and discharge, and the impact on the patient’s quality of life. Assessing the severity of the impact using the Visual Analogue Scale (VAS) for pain severity, and the Dermatology Life Quality Index (DLQI) is recommended.1 Examining the affected areas and documenting the presence of HS lesions is important so there is a record of the extent of disease prior to treatment.
The Hurley staging system is the most commonly used tool for assessing baseline disease severity in HS, but because it relies heavily on the presence of scarring it is less useful for monitoring the response to treatment. Lesion counts or a change in the number of flares of the condition are the most appropriate measures for follow-up assessments, alongside pain and quality of life scoring. The Hurley staging system defines HS disease severity as follows:1
- stage I (mild)—isolated lesions with no sinus tract formation and minimal or no scarring
- stage II (moderate)—recurrent lesions separated by normal skin with sinus tract formation and scarring
- stage III (severe)—multiple lesions coalescing into inflammatory plaques involving most of the affected region.
Assessment of patients with a new diagnosis of HS should also address the common co-morbidities of the disease:
- enquire about smoking status
- screen for cardiovascular risk factors, including:
- body mass index (BMI)
- waist measurement
- blood pressure
- HbA1c
- lipids
- assess mood using standardised depression and anxiety screening questions, such as those used in the monitoring of other chronic diseases.
Management
All patients will benefit from signposting to patient information and organisations such as the HS Trust, which offer support and advice to people with HS and their carers.1
Patients should be advised that smoking and having a raised BMI (>30 kg/m2) have both been linked with a more severe course for the disease, and offered referral to smoking cessation and weight management services where appropriate. If risk factors for cardiovascular disease are present they can be managed using the standard protocols, including advice on lifestyle factors. Persistent bowel symptoms should be investigated appropriately to exclude inflammatory bowel disease.1
If depression or anxiety have been found during the assessment, their effective treatment is important to improve treatment outcomes and quality of life.1 Occupational issues can arise due to the symptoms of HS, so giving information about employment rights may be helpful.
Initial Management of Hidradenitis Suppurativa
Most of the commonly used treatments in HS are prescribed off-licence in the UK; the only licensed treatment currently (August 2019) available for HS is the biologic adalimumab.16 The evidence base for current therapies is limited, and the BAD guideline has made recommendations based on the available low-to-medium quality evidence together with expert opinion.1
The initial management of HS depends on the severity of disease identified. If severe (Hurley Stage III) disease is found at first presentation, then referral to dermatology should be considered, as management is likely to be complex and involve both medical and surgical interventions. In mild-to-moderate disease (Hurley Stage I–II), initial management can be appropriately carried out in primary care (see Patient management pathway—hidradenitis suppurativa).1 Topical clindamycin 1% gel, applied twice daily to affected sites, has been shown to have similar effectiveness to oral tetracyclines in early, limited HS where there is minimal scarring.17 Where there is more extensive disease, scarring, or lack of response to topical clindamycin, then lymecycline 408 mg daily or doxycycline 100 mg once or twice daily, for 12 weeks, are recommended on the basis of expert opinion. Patients should be reviewed at 12 weeks, and treatment success assessed by number of inflamed lesions, pain score, and quality of life. Treatment should be continued where beneficial, as HS is a chronic disease. Treatment breaks should be used to assess the ongoing need for therapy and reduce the risk of bacterial resistance developing; the frequency and length of these breaks depends on doctor and patient preference. The provision of dressings suitable for the individual patient’s needs should also be discussed, especially if lesions are discharging, and expert opinion is that low-adherence ‘gentle border’ dressings are often most suitable for moist flexural sites.
Pain is one of the most disabling features of HS and should be managed appropriately to improve quality of life and social functioning.18 Severe pain may occur due to abscesses in acute flares. Chronic pain in HS is well recognised and thought to have a variety of mechanisms:19 it should be managed according to existing guidance on chronic pain.20
GPs are also well placed to advise on appropriate contraception, which is important in women of child-bearing age, because less androgenic contraceptive methods may benefit patients with HS,21 and many of the drugs used in HS should be avoided during pregnancy.
Referral
Where there has been an inadequate response to initial management, or if HS is severe at initial presentation, then referral to dermatology is appropriate. Some localities have a specialist clinic for HS, and management by a multidisciplinary team is recommended for more severe cases, especially if surgical input is required. For patients whose HS has not responded to a 12-week course of oral tetracyclines, or if the disease is severe, the BAD guideline recommends combination treatment with clindamycin 300 mg twice daily and rifampicin 300 mg twice daily for 10–12 weeks.1 Renal and hepatic function should be monitored in accordance with manufacturers’ advice, and patients should be advised that rifampicin reduces the effectiveness of hormonal contraception.22 If successful, this treatment can be repeated intermittently.
If there is a poor response or rapid recurrence of active disease after rifampicin and clindamycin, options include off-licence use of dapsone,1 which has shown benefits in some people with mild-to-moderate HS, although ongoing blood monitoring is required as haematological disorders are a recognised side-effect at high doses. Acitretin can also be a useful therapy for HS in men and non-fertile women, but its prolonged teratogenic effects make it unsuitable for women of child-bearing age.1 Metformin is one of the few treatments for HS that is safe in pregnancy, and may also be beneficial in people with HS who have type 2 diabetes or polycystic ovarian syndrome. The use of isotretinoin in HS is not supported by available evidence, unless there is co-existing severe acne.1
In patients with moderate-to-severe HS which has not responded to standard medical therapies, the biologic drug adalimumab has now been approved for use by NICE.23 The place of adalimumab in managing HS is determined by the severity of the disease and individual patient preference. It is used at twice the dose recommended in psoriasis (40 mg weekly for HS, rather than 40 mg alternate weeks for psoriasis) as higher doses have better outcomes in HS.23 The response to adalimumab is reviewed at 12 weeks, and if there is at least a 25% improvement in the inflammatory lesion count, with no worsening of other features such as sinuses, then it can be continued long term. Patients whose disease does not respond to adalimumab may be considered for infliximab treatment, although there is a poorer evidence base for this.23
Surgical Treatment for Hidradenitis Suppurativa
Surgical treatment of HS has a place as part of a coordinated approach to the management of the disease. Repeated incision and drainage of acute lesions as a sole treatment is widely acknowledged as ineffective, but there may be a place for minimally invasive surgery for persisting sinuses in combination with medical treatment.24 The limited evidence on surgical interventions suggests that extensive excision of all the apocrine-bearing skin at an affected site has the best chance of reducing recurrence where medical treatment has failed to control the disease, but this is major surgery with prolonged recovery times.25
Summary
Hidradenitis suppurativa is a relatively common skin condition in adults of working age, affecting women more than men, and often having a significant negative effect on mood, personal relationships, and employment spanning decades of the patient’s life. Recent evidence shows it is also associated with several chronic diseases including diabetes and the metabolic syndrome, which cause increased morbidity and mortality. Recognition by primary care clinicians of the characteristic features of HS in its early stages allows interventions to limit scarring and progression to severe disease, and also management of lifestyle factors associated with common co-morbidities. The BAD guideline aims to give clear guidance to generalist clinicians to improve the management of this distressing condition and achieve better outcomes for patients.
Dr Fiona Collier
Locum GP and GPwSI Dermatology, Clackmannanshire
Member of the BAD hidradenitis suppurativa guideline development group
| Key Points |
|---|
HS=hidradenitis suppurativa |
| Implementation actions for STPs and ICSs |
|---|
written by Dr David Jenner, GP, Cullompton, Devon The following implementation actions are designed to support STPs and ICSs with the challenges involved with implementing new guidance at a system level. Our aim is to help you consider how to deliver improvements to healthcare within the available resources.
STP=sustainability and transformation partnership; ICS=integrated care system; HS=hidradenitis suppurativa; MDT=multidisciplinary team |

